IBD-RESPONSE: Defining microbial predictors of responsiveness to biologic therapies in Crohn’s disease and ulcerative colitis
We are inviting you to take part in this study as you are soon to start, or likely soon to start a biologic (an injectable medication used to control inflammation in inflammatory bowel disease e.g. infliximab, adalimumab, vedolizumab or ustekinumab), a JAK inhibitor medication (tofacitinib) or other advanced therapy medication as treatment for Crohn’s disease (‘Crohn’s’) or ulcerative colitis (‘colitis’).
There will be no changes in the care and treatment you receive, no extra visits to hospital, and we will only collect information about how you respond to your usual treatment.
As you have been diagnosed with IBD, you have been invited to be part of the IBD-RESPONSE/ CD-metaRESPONSE sub-study.
For CDmetaRESPONSE participants, you will complete the same questionnaires and collect the same samples as participants in IBD-RESPONSE, but we will also ask you to collect your stool sample in one additional tube and complete a 4-day food diary around the time of collecting your stool sample. These additional actions will occur before you start your treatment (baseline visit) and approximately 14 weeks later.
The majority of this study, including the consent form and questionnaires can be done on a mobile device at home.
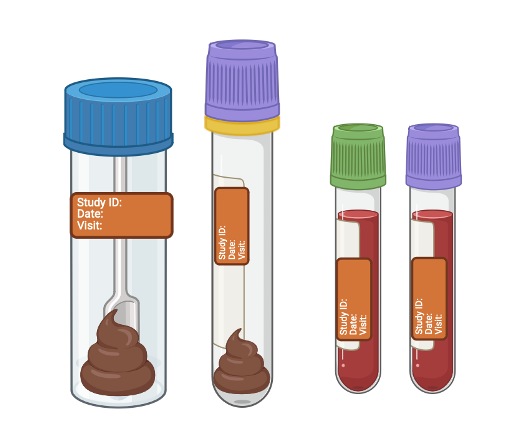
The stool samples can also be collected at home and sent back to us in the post. Bloods will only be collected if you visit the hospital for your routine care appointment.

Stool samples are collected at three time points:
(1) before you start your new treatment,
(2) around 14 weeks after starting treatment, and
(3) around 54 weeks after starting treatment.
Blood samples are collected at two time points:
(1) before you start your new treatment, and
(2) around 14 weeks after starting treatment.